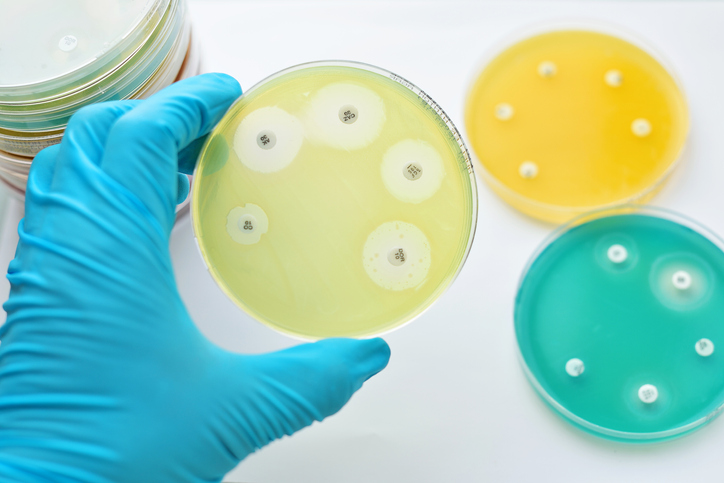
 Bacteria are very adaptive microbes. They can change their own structure to avoid being killed off by an antibiotic. Even more impressive, they can transfer that skill to other bacteria nearby by sharing DNA. These capabilities cause antibiotic resistance — the ability of bacteria to become less vulnerable or even completely immune to the medication designed to kill it. The more antibiotics we use, the more resistant they become.
Bacteria are very adaptive microbes. They can change their own structure to avoid being killed off by an antibiotic. Even more impressive, they can transfer that skill to other bacteria nearby by sharing DNA. These capabilities cause antibiotic resistance — the ability of bacteria to become less vulnerable or even completely immune to the medication designed to kill it. The more antibiotics we use, the more resistant they become.
The World Health Organization (WHO) recently identified antibiotic resistance as one of the greatest threats to global health and food security. The United Nations was so concerned that they established the Interagency Coordination Group on Antimicrobial Resistance (IACG)to combat the dire risk to human health if antibiotics become obsolete, as many scientists are predicting.
 Exposure to resistant bacteria is very difficult to avoid. The products of wastewater treatment introduced back into the water supply have been found to contain traces of antibiotic resistant bacteria. Scientists published research showing that even small concentrations of just one type of antibiotic can cause resistance to many other classes of antibiotics, further spreading resistant bacterial infections to the population.
Exposure to resistant bacteria is very difficult to avoid. The products of wastewater treatment introduced back into the water supply have been found to contain traces of antibiotic resistant bacteria. Scientists published research showing that even small concentrations of just one type of antibiotic can cause resistance to many other classes of antibiotics, further spreading resistant bacterial infections to the population.
This is not just happening in America. In Thailand, with a population of under 65 million people, antibiotic resistance causes over 38,000 deaths a year. In order to solve this global crisis, it is important to take a hard look at how the dilemma began.
Greed played a major role in spoiling antibiotics’ effectiveness
 Most people tend to blame our drug resistance problem on doctors for over-prescribing, or on consumers who don’t finish their entire course, however, antibiotics prescriptions decreased significantly following a CDC campaign in 1995. Physicians slowed their prescriptions as the public became more aware that antibiotics are not a cure-all, having no effect on viral illnesses like the flu or the common cold. In addition, the WHO has declared that shorter courses of antibiotics can be just as effective at treating many infections, hence negating the prevailing thought that not finishing a full 7 or 10 day regimen will create resistance. These interventions have had minimal impact on the problem, however, as most people are not exposed to antibiotics or resistant bacteria in this direct manner.
Most people tend to blame our drug resistance problem on doctors for over-prescribing, or on consumers who don’t finish their entire course, however, antibiotics prescriptions decreased significantly following a CDC campaign in 1995. Physicians slowed their prescriptions as the public became more aware that antibiotics are not a cure-all, having no effect on viral illnesses like the flu or the common cold. In addition, the WHO has declared that shorter courses of antibiotics can be just as effective at treating many infections, hence negating the prevailing thought that not finishing a full 7 or 10 day regimen will create resistance. These interventions have had minimal impact on the problem, however, as most people are not exposed to antibiotics or resistant bacteria in this direct manner.
Eighty percent of antibiotics sold in this country are purchased for use in animal agriculture, with 70% of those coming from classes of antibiotics that have a direct impact on human antibiotic resistance. Pumping massive amounts of vital medication into animals that Americans consume is not only risky, but a potential threat to medicine as we know it. When bacteria become resistant in animals due to overuse, humans can contract these infections by ingesting undercooked food, direct contact with animals and manure, or by simply encountering surfaces that contaminated food has touched.
 It’s not just animals anymore, either. Just this year, Florida got the ok from the EPA to spray two antibiotics commonly used in humans on about a half-a-million acres of oranges. While this practice had been approved in 2016 to be used only on an emergency basis, citrus farmers have never had the green-light to spray such large amounts of antibiotics over such large areas. The purpose: to save these oranges from an infection called “citrus greening”, which hit the US orange population in 2005 and spread quickly, affecting almost all American citrus farms by 2008. The problem: there is no cure for citrus greening. Spraying them with antibiotics has anecdotally slowed the spread of this disease, however, we are currently amid an experiment that could wipe out the effectiveness of an entire class of life-saving antibiotics.
It’s not just animals anymore, either. Just this year, Florida got the ok from the EPA to spray two antibiotics commonly used in humans on about a half-a-million acres of oranges. While this practice had been approved in 2016 to be used only on an emergency basis, citrus farmers have never had the green-light to spray such large amounts of antibiotics over such large areas. The purpose: to save these oranges from an infection called “citrus greening”, which hit the US orange population in 2005 and spread quickly, affecting almost all American citrus farms by 2008. The problem: there is no cure for citrus greening. Spraying them with antibiotics has anecdotally slowed the spread of this disease, however, we are currently amid an experiment that could wipe out the effectiveness of an entire class of life-saving antibiotics.
The alternative to antibiotic use is labor-intensive, requiring the vigilant removal of any affected produce immediately. It is often cheaper and faster to simply spray everything with antibiotics, a practice that both the CDC and the FDA opposed.
 Antibiotic use in agriculture is sometimes necessary. When a specific outbreak of disease is noted in farm animals, treatment with antibiotics is often an important intervention to prevent the spread of devastating disease in livestock. Unfortunately, most antibiotic use in chicken, beef, and pork is not used as a treatment, but as a method to increase profits. A 2012 study showed that 88% of growing American pigs were given small amounts of antibiotics in their feed, not because they were sick, but because doing so increased growth and prevented any infection that could cost the swine industry money.
Antibiotic use in agriculture is sometimes necessary. When a specific outbreak of disease is noted in farm animals, treatment with antibiotics is often an important intervention to prevent the spread of devastating disease in livestock. Unfortunately, most antibiotic use in chicken, beef, and pork is not used as a treatment, but as a method to increase profits. A 2012 study showed that 88% of growing American pigs were given small amounts of antibiotics in their feed, not because they were sick, but because doing so increased growth and prevented any infection that could cost the swine industry money.
Feeding sub-therapeutic doses of antibiotics to livestock has been knownto help them grow larger, faster, and more cheaply since the 1940’s. Research clearly showed over 40 years ago that antibiotic use in animals leads to antibiotic resistance in humans, yet very little has been done in the United States to curtail this dangerous practice that serves only to benefit big business. Greed has undoubtedly trumped health when it comes to spreading antibiotic resistant bacteria to our population.
Big Pharma is not interested in fixing the problem
 The makers of these antibiotics are far from innocent. While pharmaceutical companies once put dollars and manpower into researching and developing new classes of antibiotics, that line of product growth has slowed to a snail’s pace. The CDC says that 23,000 people in the United States die from antibiotic resistant infections yearly, and more than 2 million develop a resistant infection that is difficult and expensive to treat.
The makers of these antibiotics are far from innocent. While pharmaceutical companies once put dollars and manpower into researching and developing new classes of antibiotics, that line of product growth has slowed to a snail’s pace. The CDC says that 23,000 people in the United States die from antibiotic resistant infections yearly, and more than 2 million develop a resistant infection that is difficult and expensive to treat.
Doctors clearly need more treatment options as those numbers continue to grow. Unfortunately, only 9 new antibiotics were developed between 2005 and 2015, compared to the 270 that were created between the 1950’s and the 1970’s. By the early 1990s, about half of drug makers got out of the business of antibiotics altogether, and today, only 6 of the top 50 pharmaceutical companies are even trying to discover new antibiotics.
Shahriar Mobashery, a biochemist who is an expert in antibiotic use and drug resistance at Notre Dame, commented,
“Companies are very much interested in disease that can be managed — chronic ailments like high blood pressure…after a certain age, you would use their product till the end of your life.”
 His point of view is supported by the numbers. A 2014 presentation given by Eastern Research Group showed that while sales of antibiotics to treat urinary tract infections netted about $22 million, some classes of antibiotics, such as those used for dental procedures, actually lost money — to the tune of $4 million. In comparison, drugs to treat life-long musculoskeletal disorders (especial pain related issues) raked in around $1 billion. It shouldn’t be a surprise that almost all the top pharmaceutical companies stopped making new antibiotics years ago, citing high R&D costs and low returns on their investment. Drug companies are, indeed, companies, with the goal of generating revenue by finding the next big money-maker.
His point of view is supported by the numbers. A 2014 presentation given by Eastern Research Group showed that while sales of antibiotics to treat urinary tract infections netted about $22 million, some classes of antibiotics, such as those used for dental procedures, actually lost money — to the tune of $4 million. In comparison, drugs to treat life-long musculoskeletal disorders (especial pain related issues) raked in around $1 billion. It shouldn’t be a surprise that almost all the top pharmaceutical companies stopped making new antibiotics years ago, citing high R&D costs and low returns on their investment. Drug companies are, indeed, companies, with the goal of generating revenue by finding the next big money-maker.
With so many antibiotics becoming ineffective at such a rapid pace, their chances of striking it rich with these critical infection-fighters are slim. Therefore, there are no antibiotics available to fight against the Hospital Associated Infections (HAI’s) that are responsible for most deaths associated with antibiotic resistance. Once again, industry greed eclipses human health.
Government isn’t helping either
The lack of government response to this health crisis is disturbing to say the least. As far back as 1981, J L Armstrong et al found that water treatment plants produced a supply of water that selected for resistant organisms as compared to raw water, showing that resistant bacteria could be spread through the water supply, especially after treatment. Yet, America has been notably slow with regulation aimed at confronting this problem. The same is true of our inaction regarding livestock.
In 1986, Sweden recognized the dangers of overuse of antibiotics in farm animals, and completely banned the practice of using them for the sole purpose of growth promotion. In the 1990s, Denmark began restricting antibiotic use in animals, eventually banning them there as well. By 2006, the European Union followed suit.
 The United States, however, continues to maintain its stance that not enough proof of benefit exists to institute the same changes. While the FDA has restricted the use of some classes of antibiotics, the Environmental Protection Agency (EPA) continues to move slowly in its regulatory measures, with most of their recommendations remaining as a ‘voluntary’ phase out. The EPA is hesitant to acknowledge that any ban on farm animal antibiotic use will slow down antibiotic resistance, even though a WHO study showed that the Danish ban reduced risk to human health without significantly affecting animal health or farmers’ profits.
The United States, however, continues to maintain its stance that not enough proof of benefit exists to institute the same changes. While the FDA has restricted the use of some classes of antibiotics, the Environmental Protection Agency (EPA) continues to move slowly in its regulatory measures, with most of their recommendations remaining as a ‘voluntary’ phase out. The EPA is hesitant to acknowledge that any ban on farm animal antibiotic use will slow down antibiotic resistance, even though a WHO study showed that the Danish ban reduced risk to human health without significantly affecting animal health or farmers’ profits.
The EPA’s track record on instituting regulation to improve human health has been sluggish at best. Despite the WHO declaring that glyphosate, the most widely used herbicide in the U.S., was a probable human carcinogen (and multiple European countries banning its use) the EPA still maintains on its website that glyphosate poses no risk to human health from current uses, and that there is no evidence it causes cancer. Chlorpyrifos is the most widely used pesticide in this country but has been banned in many others. This chemical was introduced as a replacement for DDT, but subsequently was shown to cause neurological development issues in children.
Despite the American Academy of Pediatrics and the EPA’s own scientists pushing for a ban of the chemical, the EPA ignored its own research and refused to pull the trigger on a nationwide ban. Some have sited political motives as the reason, especially since the maker of chlorpyrifos, Dow Chemical, spent $13.6 million in 2016 and $5.2 million in the first quarter of 2017 on lobbying. The acting EPA Administrator even met with the CEO of Dow before he made his decision to allow the pesticide to remain in use.
This is concerning given the fact that new research shows increasing pesticide and herbicide resistance contributes to increasing antibiotic resistance. Bacteria are a part of every human being, and when consistently exposed to herbicides or pesticides, the speed at which resistance to antibiotics develops can be exponentially increased. As agriculture searches for new ways to outsmart pests and increase yields, unbiased research on the safety of new products continues to take a backseat to the bottom line.
 In another questionable call, the agency has permitted sewage sludge, also called biosolids, to be stockpiled or spread on American soil, despite warnings that this method of dealing with waste could threaten human health. The treated sludge is full of nutrients, making it a perfect (and very cheap) option for farmers to use as fertilizer, but it also contains hundreds of other substances, pharmaceuticals included. The rich soil is a breeding ground for bacteria, which, when combined with even trace amounts of antibiotics, creates the ultimate environment for antibiotic resistance to flourish. In 2018, the EPA’s inspector general statedthat they did not have enough data regarding biosolids to know if their use was safe or not. The current refrain continues to be “safe until proven deadly”.
In another questionable call, the agency has permitted sewage sludge, also called biosolids, to be stockpiled or spread on American soil, despite warnings that this method of dealing with waste could threaten human health. The treated sludge is full of nutrients, making it a perfect (and very cheap) option for farmers to use as fertilizer, but it also contains hundreds of other substances, pharmaceuticals included. The rich soil is a breeding ground for bacteria, which, when combined with even trace amounts of antibiotics, creates the ultimate environment for antibiotic resistance to flourish. In 2018, the EPA’s inspector general statedthat they did not have enough data regarding biosolids to know if their use was safe or not. The current refrain continues to be “safe until proven deadly”.
These dubious decisions, among others, have raised eyebrows, leading some to believe that current lobbying laws and corporate greed are working against policies intended to improve human health. Some regulatory practices may even be actively harming us. With so many nefarious influences contributing to research that supports health-related regulations in this country, it can be difficult to determine how best to avoid hazardous environmental exposures. Studies recognized by government as confirmation of safety often have conflicting research that regulators have chosen to overlook.
Sometimes accepted ‘Science’ is just plain wrong
 When Vancomycin was developed to treat resistant bacteria in 1972, it was widely recognized as the ‘ultimate antibiotic’ to which bacteria could not and would not be able to develop any resistance. This resistance-proof antibiotic remained so for less than eight years, when vancomycin resistant enterococcus (VRE) was first isolated. Unfortunately, history shows us that ‘sure things’ in medicine often turn out to have inadvertent outcomes.
When Vancomycin was developed to treat resistant bacteria in 1972, it was widely recognized as the ‘ultimate antibiotic’ to which bacteria could not and would not be able to develop any resistance. This resistance-proof antibiotic remained so for less than eight years, when vancomycin resistant enterococcus (VRE) was first isolated. Unfortunately, history shows us that ‘sure things’ in medicine often turn out to have inadvertent outcomes.
While bloodletting was an accepted treatment for at least 2000 years, and mercury (a now-known neurotoxin) was used for everything from baby teething to syphilis for centuries, the modern era of medicine sees these remedies as barbaric. More recently, opioids became the norm for treating pain, toxic chemotherapy became the standard for killing cancer cells, and antimicrobials like antibiotics and antivirals were handed out like candy to anyone complaining of a cough or cold. All of these were accepted as the standard of care in their time.
Now, immunotherapies are beginning to replace chemotherapies, alternative pain relief methods have supplanted rampant opioid scripts, and fecal transplants are starting to become accepted therapies as scientists come to understand the importance of the human microbiome.
It is imperative to gain perspective on current trends in treatment and prevention of disease as we move into a new era where most common antibiotics will become obsolete.
Reality Check
 In truth, commercial antibiotic use has only been around for about 80 years. Compare that to the roughly 200,000 years that humans managed to survive without them, and it might seem a little premature to sound the apocalypse alarm just yet. Although the Black Plague, which was caused by a bacterium, wiped out roughly half of Europe’s population in the 1300’s long before any commercial antibiotics were available, that scenario is unlikely to repeat itself at that scale anytime soon.
In truth, commercial antibiotic use has only been around for about 80 years. Compare that to the roughly 200,000 years that humans managed to survive without them, and it might seem a little premature to sound the apocalypse alarm just yet. Although the Black Plague, which was caused by a bacterium, wiped out roughly half of Europe’s population in the 1300’s long before any commercial antibiotics were available, that scenario is unlikely to repeat itself at that scale anytime soon.
Better sanitation, cleaner water, and improved human hygiene have all contributed to declining deaths from bacterial infections.
Today, an educated society understands the importance of handwashing, cooking food thoroughly, and the significance of taking in vitamins and minerals essential to maintaining a healthy immune system. In fact, 21stcentury medicine is moving in the direction of harnessing the body’s own natural defenses to prevent and treat disease rather than focusing on trying to ‘outsmart’ pathogens that have been evolving for eons.
Medical treatments are forever advancing and improving. Science is never settled, and no research on medical interventions should ever be stifled, especially not for the purpose of financial gain. Greed may have triggered the current threat of a post-antibiotic world, but respectable scientists and researchers will persevere in presenting novel ideas for prevention and treatment of bacterial infections, with or without an effective antibiotic in their arsenal.


Join the Conversation